How does it work?
A diagnostics revolution. Helping to protect.
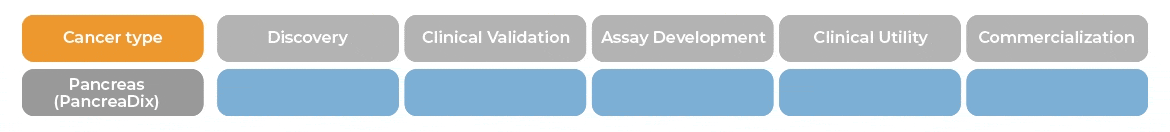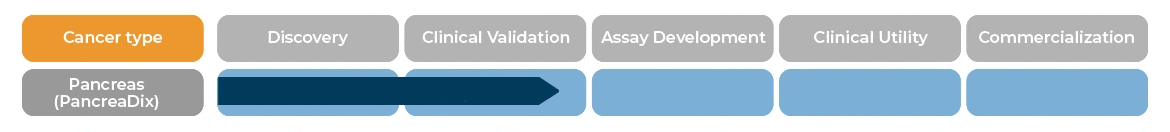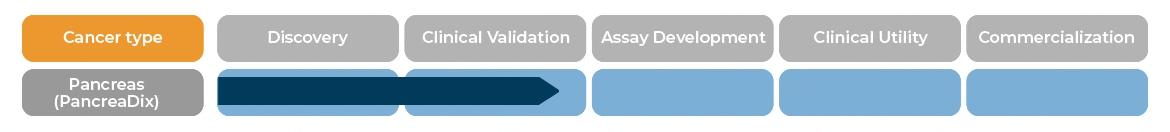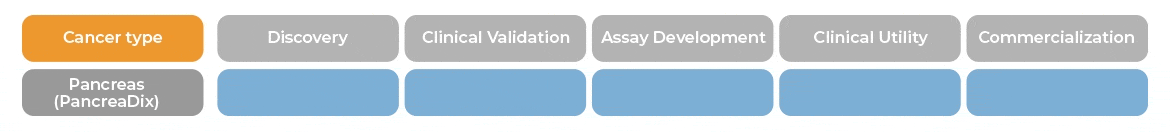
PancreaDix® identifies and measures biomarkers which are differentially expressed in blood samples of individuals with Pancreatic Cancer (PaC)
- Genetic biomarkers
- Non-invasive test in plasma
- Easy, low cost and a common technique for hospitals and laboratories
- Confirming Pancreatic cancer diagnosis, identifying benign vs. malign pancreatic cysts to avoid unnecessary surgeries and differential diagnosis between Pancreatic cancer and other pathologies.