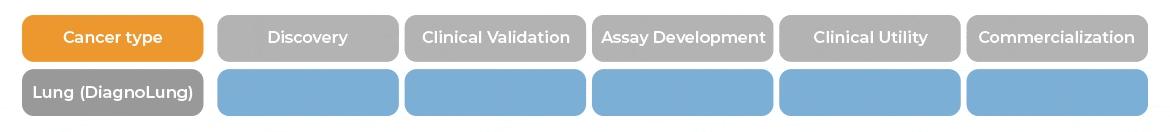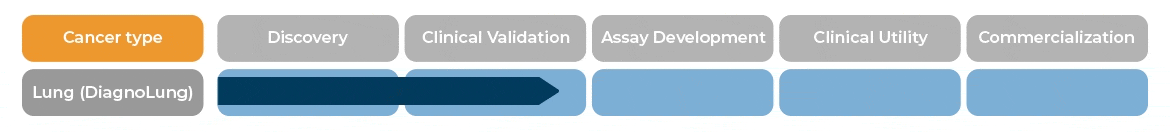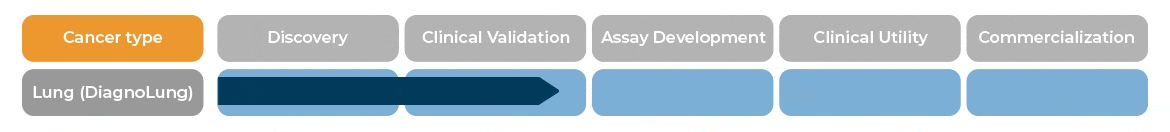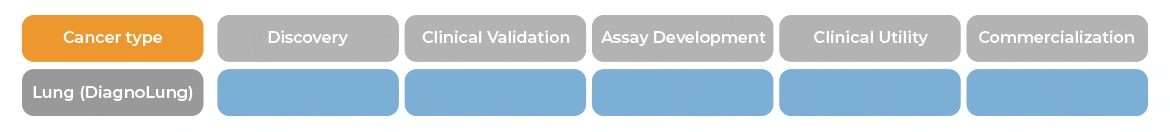
In patients with indeterminate pulmonary nodules, between 6 and 30 mm, DiagnoLung would allow physicians to identify patients with likely benign nodules who would become candidates for serial Computed Tomography (CT) monitoring within 2 years.
In a screening context, DiagnoLung is indicated to confirm positive Low Dose Computed Tomography (LDCT) results that may indicate the presence of lung cancer and should be confirmed with an invasive test (biopsies).
Performed in conjunction with a biopsy, DiagnoLung can be used to confirm cancer in patients with inconclusive cytological and histological analysis (in a clinical context, it is not possible to confirm the diagnosis in up to 50% of first bronchoscopies).
It is not a replacement for diagnostic methods or the monitoring of high-risk individuals (50 to 80 years old and in fairly good health, who currently smoke or who have quit in the past 15 years and have at least a 20 pack/year smoking history).